Hidden gems in cement
19 comments
I return! With another Science blog hehe. I promise I'm not such an intense nerd, but materials honestly fascinate me. Previously, I talked about coal, coal types and their pecific use. I'm not sure if I'm up for talking about the types of testing we had to do on them sincce that would be a breach in laboratory confidentiality, and I am definitely not ready to go to jail yet.
Anyways, aside from testing coal, I also got to experience testing cement and cement blocks from when I worked in an engineering lab. I didn't stay long there, but I did get to experience a lot of different materials there. I would say that facility-wise, it was the worst lab I've ever been to, but knowledge-wise, it was a good place to learn.
From there, I actually learned that testing cement takes about a week to complete since in that lab, we used traditional wet chemistry for it. And that involved a lot of filtering. I remember then that in a day, we had 70+ samples to dissolve, filter, dry, cool, weigh and redissolve again for the next analyte. In a day, we can mostly finish 2-3 analytes, depending on how nice management was to us that day, because on some days, they would just barge into the lab and demand us to look for this and that while in the middle of an analysis. It was annoying and we hated the management for it because we couldn't finish our backlogs because of them.

All the samples I used to analyze and more

Dissolved cement sample for sodium precipitation
Back to the analytes. In cement, you have 8 major metal oxides but for this article, I think I'll just highlight 5 since something interesting happens to those 5 components.
| Compound | Formula |
|---|---|
| Calcium Oxide | CaO |
| Silicone dioxide | SiO2 |
| ------------ | ------------ |
| Aluminum Oxide | Al2O3 |
| Iron Oxide | Fe2O3 |
| ------------ | ------------ |
| Water | H2O |
| Sulfate | SO3 |
| ------------ | ------------ |
Why am I highlighting just these components? because they are the core of cement mixes. The percentage would just depend on the type of cement they were producing (because I remember there were different kinds of cements but I forgot their specific names, i just know that calcium and sulfur levels were the only things that changed in those).
They are called core components for a reason. You see, these were the very components of Roman cement. Yes, this mixture was what built rome's great and amazingly preserved infrastructures. I dug into that and the use of limestone as concrete. It was pretty cool because What they started is still being used to this day (But with modified formulation, of course).
Back then, their concrete formulation consisted of lime stone (calcite taken from the ocean. Or they sometimes they used seashells as a replacement and that's perfectly fine because they have the same chemical constitution of Calcium carbonate (CaCO3)), then they used volcanic ash (the sulfur and alumina source) or sand (silica and alumina source) (because Rome has volcanic lands), seawater or any water then volcanic rocks as fillers.

An example of a colliseum built in rome that remained well preserved to this day
Source:
You can see how their simple formulation can make these structures last centuries. Unlike now, the concrete formulation has changed so much and the lime source has shifted to petroleum byproducts so we think it could have attributed to the compressibility weakness (But I'll talk about that on another article, maybe?). Anyways, I found something much more impressive when looking through the material and durability comparison of the Roman concrete formulation vs. The concrete formulation now. And that is the fact that Calcium in Roman concrete reacts with seawater to form aluminous tobermorite.
Aluminous Tobermite (Al-Tobermorite) is a calcium silicate crystal that kind of reinforces the concrete block and makes it stronger. Although the crystal itself isn't so strong (with a moh's hardness rating of 2.5), it still adds to the stability of the structure. It's molecular arrangement is fascinating too because it forms fine fibers and plates, And that in itself is a pretty strong structure unlike with most crystals that sometimes have holes in their structures, this one is solid (yet still soft).
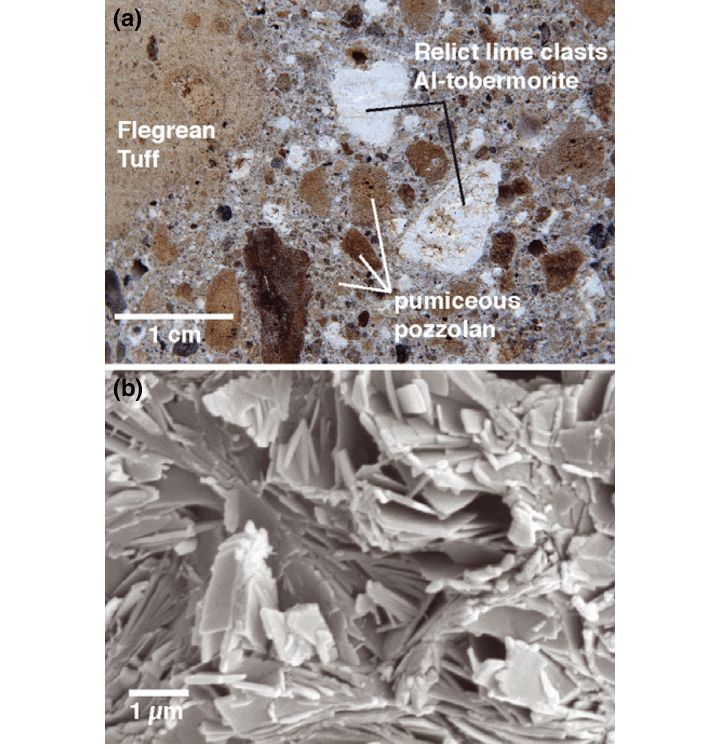
A close up of Al-Tobermite along with an SEM scan to show the crystal sheeting and foramtion
source: https://www.researchgate.net/figure/Volcanic-ash-hydrated-lime-mortar-of-the-ancient-Roman-Baianus-Sinus-concrete-late-first_fig1_251875478
Just the crystal growth that replaces the weathered parts of cement is enought to fortify the strusture, unlike now, when concrete gets soaked in saltwater, it degrades, crumbles and just adds micropollutants to the ocean. But I think this discovery of theirs worked to their advantage becaause the roman empire was surrounded with seawater and volcanic lands. I'd say they were ingenious wih their adaptability to their available resources.
A map of Rome at it's peak. I wanted to highlight the body of water that surrounded the lands
Source:
Much more so knowing that the Romans were the first in history to have a historical record of cement use.
Anyways, this is all from me for today! I still have work tomorrow and I'm still in a dilemma with my career, I swear. It pretty much explains my inactivity these days XD
Sources
Fluid chemistry, Drilling and Completion
https://doi.org/10.1016/B978-0-12-822721-3.00008-3



Comments